IPA(Isopropanol ) Dehydration
Isopropanol (IPA) is an important organic solvent widely used in the chemical, pharmaceutical, and electronics industries. In many industrial processes, IPA needs to be dehydrated and purified to meet the requirements of solvent recovery and high-purity applications. However, IPA dehydration is not a simple distillation task, but a typical azeotropic separation challenge.
IPA and water form a minimum-boiling azeotrope under atmospheric pressure, with an azeotropic composition of about 87.4–87.8 wt% IPA and a boiling point of about 80.3–80.4 °C. This means that when the mixture approaches the azeotropic point, the separation efficiency of conventional distillation drops significantly, making it difficult to obtain high-purity IPA by ordinary distillation alone. As a result, IPA dehydration often requires enhanced separation methods such as azeotropic distillation, extractive distillation, adsorption, or membrane-based processes.
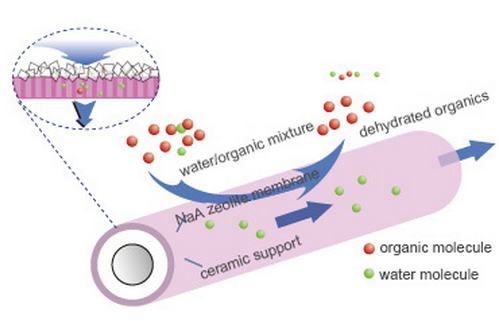
Pervaporation provides an efficient solution for IPA dehydration. In this process, the feed enters the membrane system in the liquid phase, and separation is achieved through the differences in solubility and diffusion rates of the components within the membrane. For IPA dehydration, hydrophilic membranes are typically used to preferentially remove water. The permeating component is then removed from the permeate side in vapor form, usually by vacuum or sweeping gas, allowing the water content in the retained IPA stream to be continuously reduced.
In industrial applications, pervaporation membranes are usually not intended to replace the upstream distillation section completely. Instead, they are more commonly used as a final dehydration unit after distillation. The upstream distillation first concentrates IPA to near the azeotropic composition, and the downstream pervaporation membrane then removes the remaining water to achieve higher product purity. This hybrid process is widely recognized as an effective route for IPA dehydration.
From an engineering point of view, the value of pervaporation membrane in IPA dehydration is mainly reflected in the following aspects:
Overcoming the azeotropic limitation
Conventional distillation reaches a clear bottleneck near the IPA–water azeotrope, while pervaporation can continue removing water and further increase IPA purity.
Suitable for deep dehydration
When the water content becomes low, removing trace water by distillation usually requires high energy input. Pervaporation is more suitable for this final dehydration stage because separation is achieved through selective membrane transport.
Easy process integration
By combining distillation + pervaporation, the overall process can be made more compact and efficient, while reducing the burden on complex dehydration sections. Economic comparisons in the literature specifically evaluate this hybrid route for IPA/water dehydration against conventional azeotropic distillation.
Good operational flexibility
The dehydration performance can be adjusted through parameters such as membrane area, feed temperature, feed composition, and permeate-side pressure. Studies on IPA/water pervaporation show that operating conditions strongly influence flux, membrane area requirements, and overall process design.
Overall, IPA dehydration is one of the important applications of pervaporation membrane technology in solvent purification and recovery. For IPA–water systems near the azeotropic composition, pervaporation provides an efficient deep dehydration solution and offers a practical technical route for high-purity IPA production, solvent recycling, and hybrid process intensification. With the continuous development of membrane materials and system integration technologies, pervaporation membranes are expected to show even greater value in IPA dehydration applications.